A doctor from Chhindwara district in Madhya Pradesh has been arrested after prescribing a toxic cough syrup that allegedly led to the deaths of 11 children. The incident has triggered nationwide outrage and prompted the state government to take swift legal and administrative action against those responsible, including the manufacturer of the syrup.
Doctor Arrested After Tragic Deaths in Chhindwara
The arrested doctor has been identified as Dr. Praveen Soni, a pediatrician practicing in Parasia town of Chhindwara district. According to officials, most of the affected children were treated at Soni’s private clinic. Despite being a government doctor, he had been seeing patients privately and allegedly prescribed Coldrif syrup, which has now been found to contain a deadly chemical.

Table of Contents
The tragic sequence began when several children who had been given the syrup developed serious health complications, including kidney failure. Eleven children eventually lost their lives, leading to widespread shock and grief among families in the region.
Coldrif Syrup Found Contaminated with Deadly Chemical
Laboratory analysis confirmed that Coldrif cough syrup, manufactured by Srisan Pharmaceuticals based in Kanchipuram, Tamil Nadu, was contaminated with 48.6% diethylene glycol — a highly toxic industrial solvent. Diethylene glycol is not meant for human consumption and can cause acute kidney damage, neurological disorders, and even death if ingested.
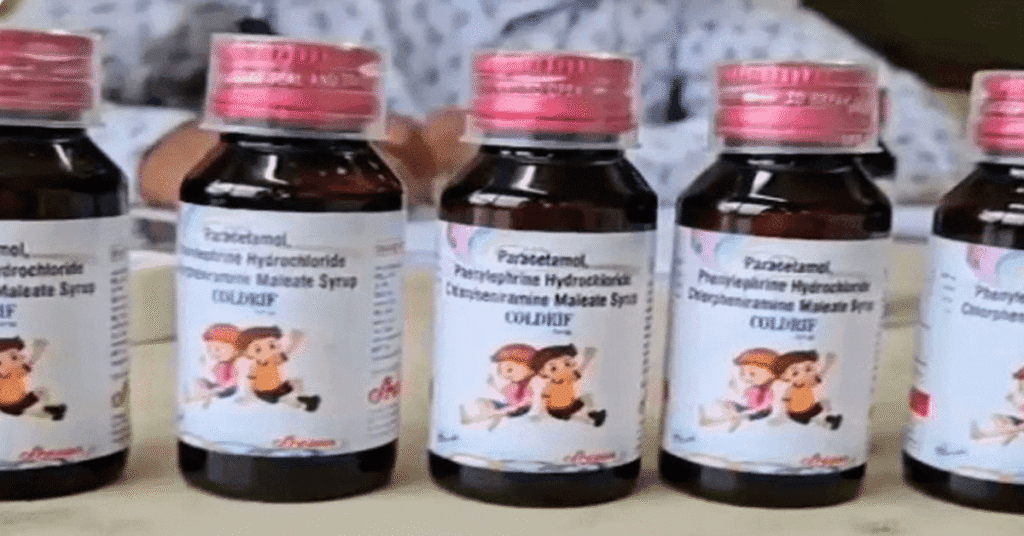
The Tamil Nadu Directorate of Drug Control declared the product “Not of Standard Quality” after receiving test results from the Drug Testing Laboratory in Chennai. Following this, the Madhya Pradesh government immediately banned the sale of Coldrif syrup across the state as a precautionary measure.
Second Cough Syrup Also Under Investigation
In addition to Coldrif, local authorities have suspended the sale of another cough syrup called ‘Nextro-DS’, pending laboratory results. Officials said that while the Coldrif test report confirmed toxic contamination, the Nextro-DS test report is still awaited.
The contamination came to light after families reported that their children initially showed mild symptoms — such as fever and cough — in early September. They were prescribed the syrup as part of routine treatment. However, within days, their condition worsened drastically. The children experienced reduced urine output, kidney infections, and multiple organ failure.
Post-mortem and biopsy results later revealed the presence of diethylene glycol in their systems, confirming the syrup as the cause of death.
Government Crackdown on Manufacturer and Distributors
The Madhya Pradesh government has filed a criminal case against Srisan Pharmaceuticals, the company responsible for producing Coldrif syrup. Authorities are now investigating the supply chain, including distributors, chemists, and local suppliers, to determine how the contaminated batch reached patients in Chhindwara.
State health officials confirmed that all remaining stock of Coldrif and Nextro-DS has been seized from medical stores in the affected districts. A state-level investigation team has also been formed to monitor the case and ensure accountability.
Chief Minister Promises Strict Action
Madhya Pradesh Chief Minister Mohan Yadav described the deaths as “extremely tragic” and assured that no one responsible will be spared. Taking to social media platform X (formerly Twitter), he wrote:
ALSO READ- Tata Nexon records sales in September 2025, with over 22,500 units sold
Yadav added that his government had coordinated with the Tamil Nadu government to test samples from the Kanchipuram factory and that the investigation report confirming contamination was received on Saturday. “Strict action has been taken based on the report. The guilty will not be spared at any cost,” he said.
Nationwide Concern Over Drug Safety
This incident has reignited concerns over pharmaceutical quality control in India, especially after similar tragedies in Gambia and Uzbekistan, where contaminated Indian-made cough syrups were linked to child deaths. Experts say the case underscores the urgent need for stricter regulation, frequent testing, and better oversight of drug manufacturing units across the country.
The Central Drugs Standard Control Organization (CDSCO) is expected to conduct an independent inquiry and may also initiate a nationwide inspection of other products made by Srisan Pharmaceuticals.
Families Demand Justice
Distraught families in Chhindwara continue to demand justice for their children and stronger laws to prevent such incidents in the future. Many of them said they had trusted the doctor’s prescriptions and had no idea the medicine could be dangerous.

“This shouldn’t happen to anyone else,” said a grieving parent. “We took our child to the doctor for a common cold, and now we’ve lost him forever.”
The arrest of Dr. Praveen Soni and the government’s action against Srisan Pharmaceuticals mark the beginning of a deeper probe into the systemic lapses in India’s drug regulation. As the investigation continues, the Chhindwara tragedy serves as a stark reminder of the devastating consequences of pharmaceutical negligence and the urgent need for accountability in public health.











